HIFU treatment : the care pathway

Discover the journey of care for Bill, who suffers from localized prostate cancer. After the clinical evaluation of his cancer, he discussed treatment options with his urologist. He chose non-invasive therapeutic ultrasound (
Learn more
HIFU in the news
Quality of life

Formerly considered to be incompatible with cancer, therapeutic advances and improvement in cancer survival have recently placed quality of life at the heart of patients' decision about prostate cancer management. Quality of life is akin to a person's judgment on his life and his psychic equilibrium, ...
Learn more
The Prostate
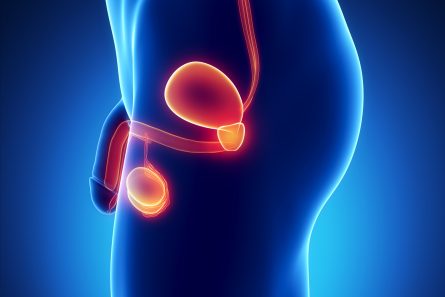
The prostate is a small gland in the male reproductive system. It is situated in front of the rectum, below the bladder, and surrounds the urethra, which is the tube through which urine flows out of the body. The main function of the chestnut-sized prostate is to secrete part of the seminal fluid that combines with sperm to form semen.
Learn moreFrequently Asked Questions
What is prostate cancer incidence in the world ?
What are the Ablatherm® HIFU and Focal One® devices?
What are my prostate cancer treatment options?
What is the HIFU treatment and under what conditions would I choose this treatment?
What are the side effects associated with the HIFU prostate cancer treatment?